
Overview of IMMS (Intelligence Manufacturing Management System)
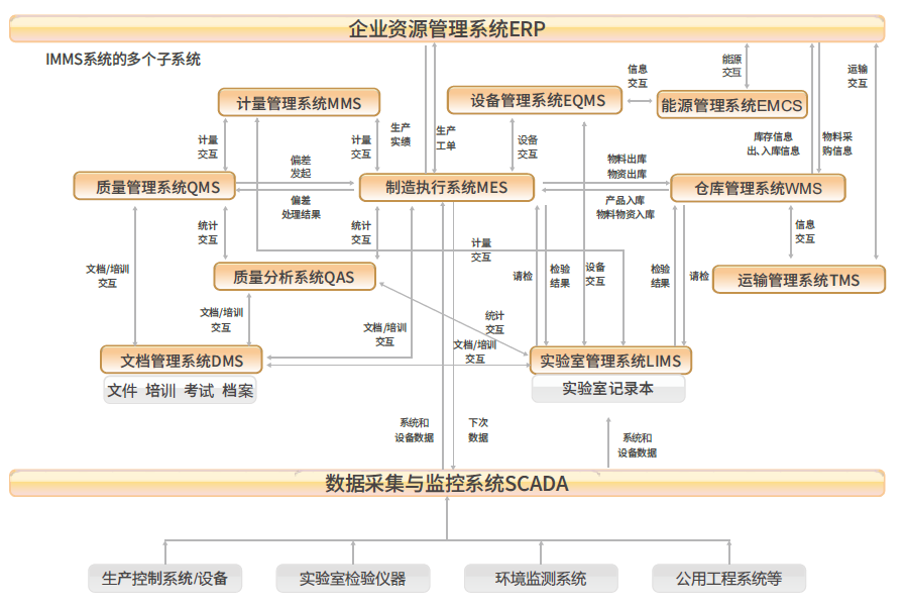
GELEG set up a professional software technical team, integrated more than 20 years of GMP consulting experience into software research and development, and created a compliant, simple, practical and efficient Intelligence Manufacturing ManagementSystem with independent intellectual property rights. IMMS). The platform includes Manufacturing Execution System (MES), Laboratory Management System (LIMS), Warehouse Management System (WMS), Document management system (DMS), Metrological Management System (MMS), Equipment Management system (EQMS), Quality Management system (QMS) and Quality analysis system (QAS), transportation management system (TMS), and data collection Subsystems such as assembly and Monitoring System (SCADA) and Energy Management System (EMCS).
IMMS intelligent system platform is committed to the realization of "giving every pill a new digital life", the platform groundbreaking integration of most of the information systems used in the production quality activities of pharmaceutical companies, with an intelligent platform to solve the production, inspection, warehousing, documentation, quality and other problems. Efficient transfer of information between modules, avoiding the development of complex system interfaces! Each system of the platform can be deployed on a server as a whole, which greatly reduces the hardware cost in the process of informatization of pharmaceutical enterprises, and also greatly reduces the cost of enterprise software procurement, verification, data docking and time costs. The platform will provide efficient and reliable intelligent manufacturing solutions for the pharmaceutical industry, helping the digital transformation and high-quality development of the pharmaceutical industry.
The multiple subsystems of IMMS, and the information transfer of each system:
The main features of IMMS intelligent management platform are as follows:

